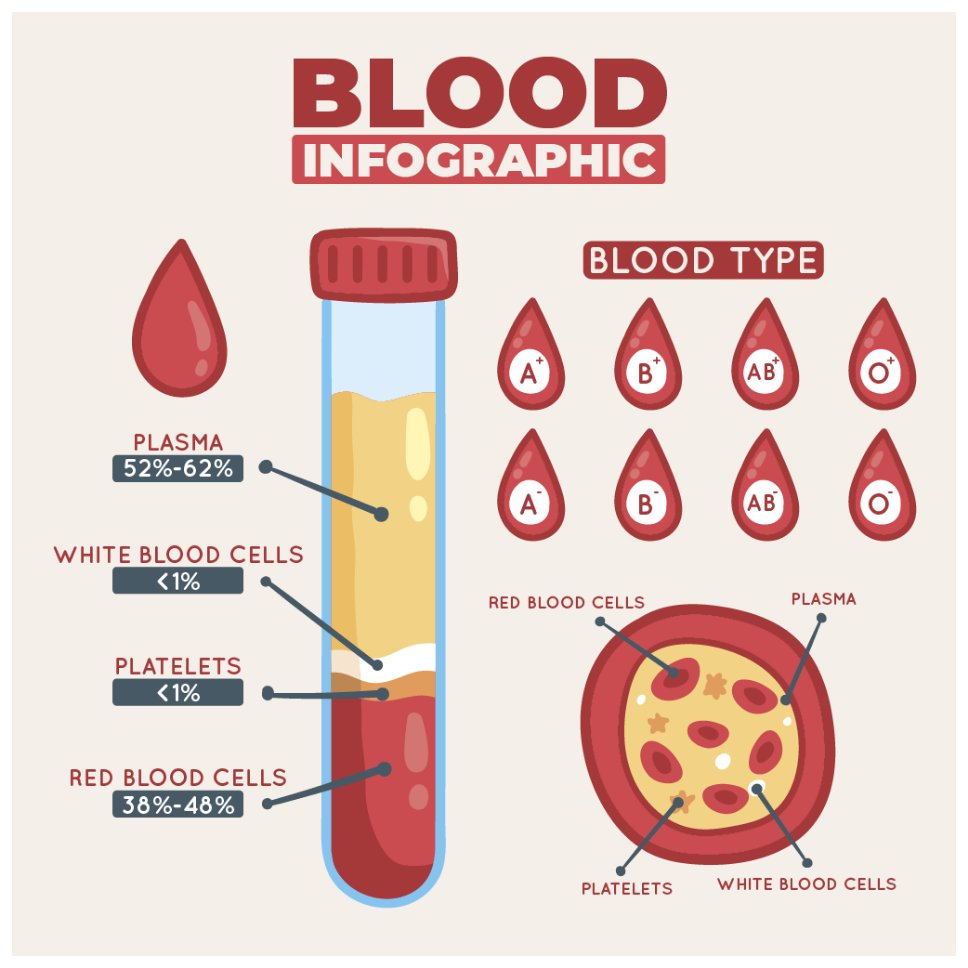
Leukaemia is a blood cancer, and its progression is assessed differently from most other cancers. Cancerous white blood cells circulate throughout the bloodstream and bone marrow from the point of diagnosis. Staging is therefore determined through blood cell counts, bone marrow findings, and organ enlargement, not tumour size. Each type of leukaemia has its own classification system.
Acute Leukaemia (ALL and AML)
Acute Lymphoblastic Leukaemia (ALL) and Acute Myeloid Leukaemia (AML) do not use a traditional numbered staging system. Instead, doctors classify the disease based on a few factors:
- The percentage of blast cells (immature cancer cells) in the blood and bone marrow.
- Genetic and chromosomal abnormalities found in the cancer cells.
- The patient's age and overall blood cell counts.
- The current disease phase: untreated, in remission, or recurrent.
The primary treatment goal for acute leukaemia is remission, a state in which no leukaemia cells are detectable in the body.
Chronic Leukaemia (CLL and CML)
Chronic leukaemia progresses more slowly and is classified using established staging systems.
Chronic Lymphocytic Leukaemia (CLL) is staged using the Rai system, ranging from Stage 0 to Stage IV:
- Stage 0: High lymphocyte count in the blood with no other symptoms.
- Stage I: Swollen lymph nodes are present.
- Stage II: The spleen or liver has become enlarged.
- Stage III: Anaemia (low red blood cell count) has developed.
- Stage IV: An excessive number of lymphocytes in the blood and platelet count has dropped, increasing bleeding risk.
Chronic Myeloid Leukaemia (CML) is classified into three phases based on the percentage of blast cells:
- Chronic Phase: Fewer than 10% blast cells. Symptoms are mild and respond well to treatment.
- Accelerated Phase: 10% to 19% blast cells. Disease progression is faster and harder to control.
- Blastic Phase: 20% or more blast cells. Requires intensive clinical care.